The Atom For Kids
When you get right down to it, everything in the world, and the universe for that matter is made up of atoms. Atoms are the basic building blocks that are used for every type of matter in the known universe.
Atoms are extremely small and are made up of even smaller particles.
Related: Try our atom quiz once you’ve read up on these atom facts!
These basic particles that make up an atom’s structure are known as electrons, protons, and neutrons.
Different substances have different numbers of these three particles, which helps to change the makeup of the matter.
Atoms fit together to create matter. It takes a ton of atoms to make up anything. This is because they are so incredibly small.
Related: Molecule facts for kids
Take the human body for example. The number of atoms in the human body is so incredibly high, it is almost beyond comprehension.
Even in the smallest person, like a baby, there are trillions upon trillions of atoms.
Depending on what type of matter you are looking at, there will be a different number of atoms. Within those same atoms, there will be differing numbers of protons, neutrons, and electrons.
Atoms can last pretty much forever. They can be changed and undergo different chemical reactions, wherein they share their electrons with other atoms.
The reason that atoms can last for such a long time is that their nucleus, or the center of the atom, is so very hard to break apart.
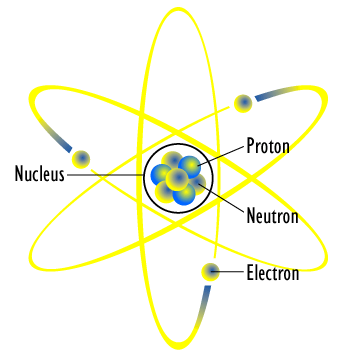
Structure of the Atom
At the center of an atom is a structure called the nucleus. Inside of this nucleus is where you will find all of the protons and neutrons.
Electrons are found outside of the nucleus and orbit around it constantly. Both electrons and protons have a charge, which is either negative or positive.
Neutrons, as you can guess by their name (‘neutral’), have no charge.
If there are more protons than electrons, then an atom is said to be positively charged. If there are more electrons than protons, then it is said to be negatively charged.
If an atom has the same number of electrons and protons, then the atom has a neutral charge.
Protons
Protons, as their name would suggest, are positively charged particles that are found within the nucleus of an atom. Every element found in nature or made in a lab has at least one proton.
Electrons
Electrons are negatively charged particles that are found outside of the nucleus of an atom. They are constantly spinning around the nucleus.
They spin at such a high rate that it can be very difficult for scientists to observe them. Electrons are the smallest particles in an atom.
They are attracted to the positive charge of the protons, which is why they orbit around the nucleus.
Neutrons
Neutrons are neither positively nor negatively charged. Instead, they are neutral in nature.
The number of neutrons within the nucleus of an atom plays a big role in determining both its mass and radioactivity.
Atom Quiz
Now, test your knowledge of the atom by taking this atom quiz!