Solids, Liquids, and Gases For Kids
Whether you are looking at elements and matter in the laboratory or in nature, there are three different types of matter that you will be able to observe.
These three are known as solids, liquids, and gases.

Take a look around you right now. Odds are, you can observe at least one of these three states of matter.
Your desk is a solid, your drink a liquid, and, of course, the air you are breathing is a gas!
States of Matter
Atoms and molecules do not change. They always have the same numbers of protons and electrons. What can change, however, is how they behave.
Depending on the state of matter that the atoms and molecules are in, they will act differently:
Solid
Solid Definition: In a solid state, molecules are packed closely together and usually arranged in a regular pattern.
This restricts the movements of the electrons until they can barely move at all. This is why solids are so hard.
Their molecules are jam-packed together so tightly that they cannot move.
Liquid
Liquid Definition: A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure.
When matter is in a liquid state, it is able to move more freely. This is why a liquid seems to flow.
The molecules are still held together, but not nearly as tightly as when they were in a solid state.
Gas
Gas Definition: Gas is a state of matter that has no fixed shape and no fixed volume.
Whenever matter becomes a gas, the molecules are so loose that they can move freely about any space that they might find themselves.
The molecules move very quickly in this state.
How Does Matter Change from One State to Another?
To put it as simply as possible, there are only two ways to change the state of matter.
You can apply or remove heat, or you can apply pressure.
For an example, let’s take a look at all the states of matter with water.

Solid
When water is a solid, it is called ice. It became ice because all of the heat was removed, which slowed down the molecules so much that it turned into a solid.
Related: Water Cycle for kids
Liquid
To change water from this solid state, you just need to apply some heat. If you set a piece of ice out on a windowsill, it will heat up.
This loosens the molecules up and allows them to move more freely as the ice melts. It then becomes a liquid, which in this case, is plain old water.
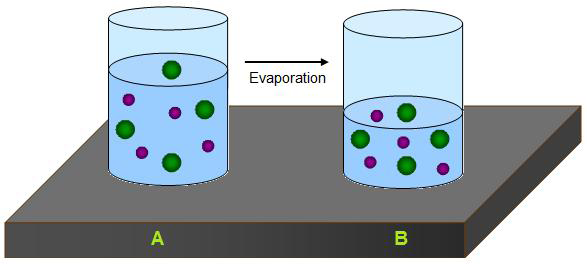
Boiling is the change of a liquid to a vapor, or gas, throughout the liquid.

Gas
If you want to change water from a liquid to gas, all you need to do is apply even more heat. As water is heated to a boil, the molecules will become even looser and start moving around more rapidly.
This will create water vapor or steam, which is water’s gaseous form.
Evaporation is the change of a substance from a liquid to a gas.
Are there any other forms of matter?
Yes, though we don’t get to observe them all that often. The first one is called plasma.
Plasma occurs at ridiculously high temperatures and can be found in stars and lightning bolts.

The other state has a bit of a fancy sounding name, which is Bose-Einstein Condensates.
This only occurs in a lab, where temperatures can be lowered a ton.

